Examine, a free weekly newsletter covering science with a sceptical, evidence-based eye, is sent every Tuesday. You’re reading an excerpt – sign up to get the whole newsletter in your inbox.
They call it beaver fever, and there’s a good chance you’ve had it. Or you might remember when millions of Sydneysiders were forced to boil their drinking water due to giardia contamination during the 1998 Sydney water crisis.
The giardia parasite infects 280 million people per year, including 600,000 Australians, by colonising the small intestine, robbing its host of nutrients and unleashing cramping, bloating and greasy diarrhoea.
The giardia parasite infects many millions of people each year, but scientists have been unsure for decades whether it reproduces sexually or not.Credit: Getty Images/Science Photo Library RF
Then it exits the body within a hardy cyst that can survive in water for months and withstand chlorine. The infection spreads when a host consumes the cyst through contaminated water or food. In this way, the parasite can jump between hosts, including between pets and their owners. (In the US, beavers carry the bug through their watery habitats, hence the nickname.)
But a huge question hangs over this notorious bug – is it a sexual being or not?
Knowing whether giardia reproduces sexually or asexually (i.e. cloning itself like bacteria) is crucial to understanding how the parasite evolves and spreads, especially because it has gained resistance to frontline drugs. About 20 per cent of cases now can’t be treated by major medicines.

A front page from The Sydney Morning Herald during the 1998 Sydney water crisis.Credit: Fairfax Media
The giardia mystery is not only imperative to public health but it is also a challenge to evolution as we know it. Yet the answer has never been clear.
Now, after 50 years, Australian-led research has solved it – and uncovered a new theory for how infections can jump from animals to humans, giving rise to new diseases called “zoonoses”.
The Red Queen and an asexual scandal

The Red Queen lectured Alice about running to stay in place – and gave rise to a key evolutionary theory.Credit: John Tenniel
In Lewis Carroll’s Through the Looking Glass, Alice sprints through a forest with the Red Queen, but realises she isn’t moving.
The queen tells Alice: “Now, here, you see, it takes all the running you can do, to keep in the same place.”
This scene inspired the Red Queen hypothesis: the idea that just to stay alive, a species has to constantly evolve in an unrelenting arms race with its competitors.
Simple single-celled organisms (prokaryotes) such as bacteria do this by quickly cloning themselves, which every so often results in an advantageous mutation. Their genomes are tiny, so they can reproduce fast.
Larger, more complex organisms (eukaryotes, like us) have a problem: their genomes are large and slow to replicate, and therefore they are vulnerable to being evolutionarily out-foxed by more nimble bacteria and viruses.
The solution? Sex.

Beavers in the US carry giardia and spread it through their watery homes – hence the nickname “beaver fever”.Credit: Ann Cameron Siegal
Recombining genes through sex means organisms can ditch bad mutations and pass on the good ones. Sex also accelerates natural selection by shuffling different genes together.
It’s so crucial to complex life that examples of eukaryotes that don’t have sex are dubbed “asexual scandals”. They don’t run fast enough to stay still, and they risk sliding backwards into evolutionary oblivion.
Another “asexual scandal” are the all-female bdelloid rotifers, although they do exchange genetic material in ways other than sex.Credit: Alamy Stock Photo
Rare examples of these scandalous virgins include oribatid mites (soil-eating bugs) and bdelloid rotifers (all-female pond-dwelling sluglike beasties).
Most of these “asexual eukaryotes” do, however, exchange genes by some mechanism that isn’t technically sex.
But giardia has remained a stubborn mystery – one of the last true asexual scandals. How could this ancient eukaryotic parasite possibly have persisted through millennia without sex, the ultimate gene-shuffling survival strategy?
Professor Aaron Jex, head of the parasite lab at Melbourne’s WEHI research centre, teamed up with international colleagues to put this evolutionary enigma to bed.
The mystery solved
Using advanced genetic screening techniques on 100 different isolates of the bug gathered from people, dogs, cats, sheep, pigs and a beaver, they revealed a fascinating answer: there were sexual and asexual types of giardia.
The reason behind decades of confusion became clear. Scientists studying the standard lab-grown version of giardia had unknowingly been testing an asexual evolutionary oddity rather than the main, sexual strain of the parasite, Jex says.

Parasite professor Aaron Jex from WEHI solved the mystery, and proposed a new theory about how diseases find new hosts.Credit: Matt Turner/WEHI
Strangely, Jex and his team found the sexual form of giardia only in people, while the asexual version had a range of animal and human hosts.
That’s weird, he says, because the sexual version of the parasite is the one you’d expect to evolve faster and adapt quickly to new hosts.
“And yet, it was our asexual parasite that was showing up in many different hosts. That didn’t make a lot of sense,” he says of the results published in Nature Communications.
“This was another piece of evidence into how zoonoses may arise.”
Professor Aaron JexThere are other examples, though, of pathogenic fungi and parasitic worms in which asexual offshoots are the ones that leap to new hosts.
In light of this, Jex and his colleagues proposed a new theory about how pathogens can jump from animals to people – the process that gave rise to the COVID-19 pandemic.
A new theory for zoonoses
Sexual parasites quickly discard bad mutations, such as tweaks that make them less infectious to their host. Sex-accelerated adaption ensures the parasite remains finely tuned to target one kind of animal.
Asexual organisms, however, can’t easily shed those unwanted mutations. As the parasites clone themselves, the same bad tweaks in DNA accumulate in the population under a process dubbed Muller’s Ratchet. That could gradually make the parasite less finely tuned to just one host.
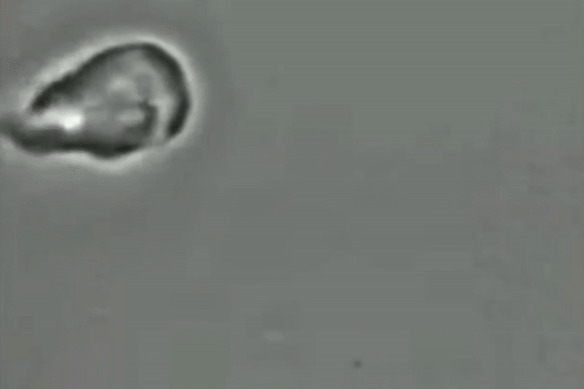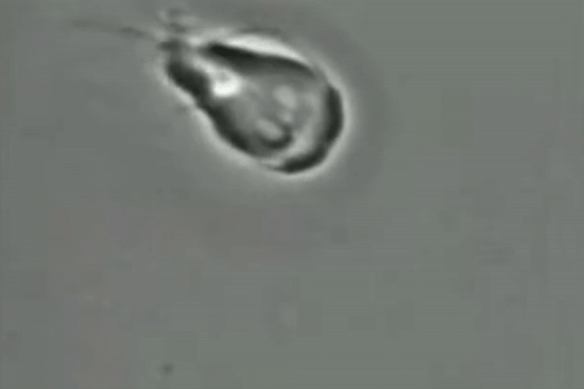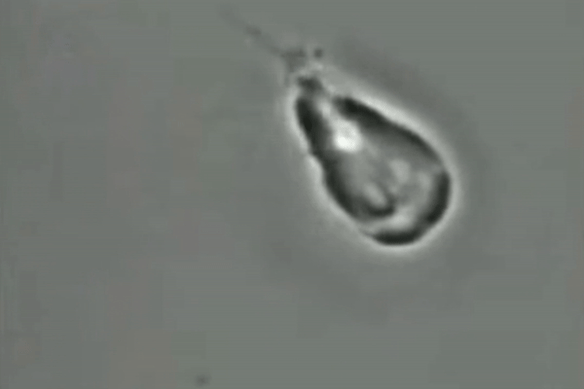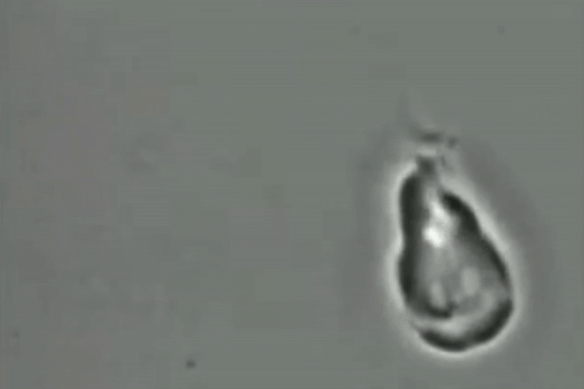
For decades, scientists have been unsure if the giardia parasite reproduces sexually or asexually.
So, the theory goes, as asexual giardia loses evolutionary fitness and host specificity, the trade-off could be that it can infect a wider range of hosts.
“Asexual lineages basically became like a tool species could use that would allow them to relax evolutionary selection and infect new hosts,” Jex says.
Loading
There may be a mechanism that then allows asexual populations to re-evolve sex, or pass genes to the sexual lineages so they can carry on in new hosts, but the idea remains theoretical, he says.
“This was another piece of evidence into how zoonoses may arise, through this process of asexual offshoots allowing the organism to relax its selective pressure to an extent where it could experiment with new mutations that would allow it to branch into new hosts, and then take off.”
The finding sheds key light on zoonoses – one of humanity’s greatest threats – and it could inform the development of drugs that better treat giardia.
And while the asexual, species-jumping type of giardia causes about half of human infection, it is ultimately doomed. The sexless lineage evolved between 600 and 2000 years ago and, since then, it has accumulated bad mutations it can’t cast off, meaning it will almost certainly die out.
This is called “mutation meltdown”, Jex says: a fate that proves the essential nature of sex, and the truth of the Red Queen’s warning.
The Examine newsletter explains and analyses science with a rigorous focus on the evidence. Sign up to get it each week.
Most Viewed in National
Loading